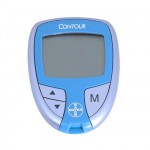
Bayer HealthCare today announced that the U.S. Food and Drug Administration (FDA) has cleared its CONTOUR® Next EZ, a new blood glucose monitoring (BGM) system for use in the United States.

Bayer HealthCare today announced that the U.S. Food and Drug Administration (FDA) has cleared its CONTOUR® Next EZ, a new blood glucose monitoring (BGM) system for use in the United States.